





Related Attributes
Product details
Etravirine, in combination with other antiretroviral agents, is indicated for the treatment of antiretroviral treatment-experienced adult patients with human immunodeficiency virus type I (HIV-1) infection.
Patients are required to have evidence of viral replication and evidence of resistance to a non-nucleoside reverse transcriptase inhibitor (NNRTI) and other antiretroviral agents.
The indication is based on an analysis of Week 48 results from 2 randomised, double-blind, placebo-controlled Phase III clinical trials.
Both trials were conducted in previously treated patients with resistance to non-nucleoside reverse transcriptase inhibitors (NNRTIs) (present at screening and/or documented in the medical record) and resistance to protease inhibitors (PIs), with the addition of this product to background therapy (BR).
The following should be considered when using this product: the patient's treatment history as well as resistance testing may guide treatment with this product.
The combination of only this product plus a nucleoside or nucleotide reverse transcriptase inhibitor is not recommended in patients with virological failure following NNRTI therapy.
This product is not recommended for use in children, adolescents, and adult patients receiving initial antiviral therapy.

Uses and functions of Etravirine.
The following should be considered when using Etravirine: the patient's treatment history as well as resistance testing may guide treatment with this product.
The combination of only this product plus a nucleoside or nucleotide reverse transcriptase inhibitor is not recommended in patients who have virological failure following NNRTI therapy. This product is not recommended for use in children, adolescents, and adult patients receiving initial antiviral therapy.

Pharmacological Effects of Etravirine.
Etravirine may be used in combination with other antiretroviral drugs in adult HIV-1-infected patients who have developed drug resistance after initial treatment with antiretroviral drugs.
Etravirine belongs to the group of non-nucleoside reverse transcriptase inhibitors (NNRTIs) of human immunodeficiency virus type 1 (HIV-1), which bind directly to the HIV-1 reverse transcriptase enzyme and block both RNA-dependent and DNA-dependent DNA polymerase activity by disrupting the enzyme's catalytic site.
Etravirine does not inhibit human alpha, beta and gamma DNA polymerases .

Production method of Etravirine Powder.
Etravirine has a high genetic barrier to resistance development. In the phase IIb trial, among multiple-treatment patients, including those infected with NNRTIs-resistant and protease inhibitor (PIs)-resistant viruses, TMC125 was active against existing NNRTIs-resistant HIV and was similarly tolerated to controls.
WHY CHOOES US?

OUR CERTIFICATE

CUSTOM PROCESS

OUR PACKAGE

OUR EXHIBITION

OUR FACTORY

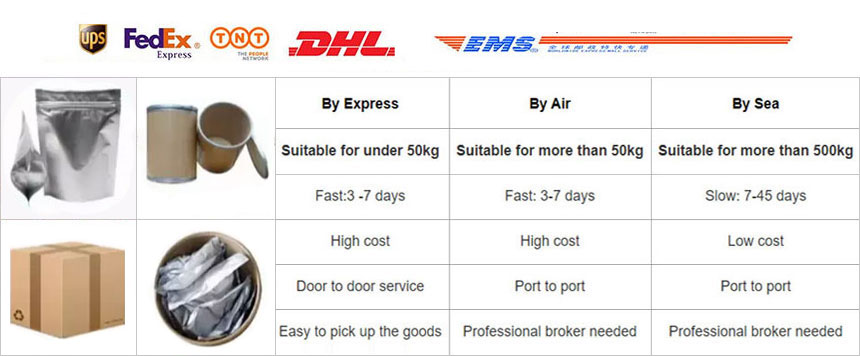
Shipping
Pharmaceutical Intermediate manufacturers
©2022 Xi'an Henrikang Biotech Co., Ltd.,