





Related Attributes
Product details
Amitriptyline inhibited trichostatin-stimulated cyclic AMP aggregation with an EC50 value of 16.2 μM in intact CHO/DOR cells.
Amitriptyline concentration-dependently stimulated ERK1/2 and GSK-3β phosphorylation with an EC50 value of 9.0 μM in CHO/DOR cells.Amitriptyline (15 μM) stimulated ERK1/2 phosphorylation in C6 cells.
Amitriptyline (30 μM) inhibited trichothecene-stimulated adenylate cyclase activity and antagonised the inhibitory effect of (-)-U50,488 in rat nucleus ambiguus.Amitriptyline binds to the extracellular structural domains of TrkA and TrkB and promotes heterodimerisation of the TrkA-TrkB receptor.
Amitriptyline (<500nM) promotes TrkA autophosphorylation in primary neurons and induces neurite proliferation in PC12 cells.Amitriptyline selectively protects T17 cells from apoptosis with an EC50 of 50nM.

Uses and functions of Amitriptyline Hydrochloride.
Amitriptyline Hydrochloride is an antidepressant with antidepressant effects and strong sedative effects. It is mainly used for endogenous exogenous depressive symptoms, menopausal depression, neurotic depression and depressive symptoms of organic psychosis, and also for enuresis in children and adolescents.

Pharmacological Effects of Amitriptyline Hydrochloride.
Amitriptyline hydrochloride is a tricyclic antidepressant that works by inhibiting the reuptake of 5-hydroxytryptamine and norepinephrine, with stronger inhibition of 5-hydroxytryptamine reuptake and stronger sedative and anticholinergic effects.

Production method of Amitriptyline Hydrochloride Powder.
Obtained from dibenzo[alpha,d]cycloheptadienone-(5) by addition, elimination and salt formation. Dried magnesium flakes and ether were put into a reaction tank and heated to reflux. The ether solution of dimethylaminochloropropane was added dropwise first, and then the ether solution of dibenzocycloheptadienone was added dropwise.
Reflow evaporate the ether, add toluene, cool and add saturated aqueous ammonium chloride solution. Stop stirring, separate the toluene solution, and then extract the aqueous layer with toluene for 5 times. Combine the toluene solution, wash with warm water 3 times, dry with anhydrous sodium sulfate, filter.
The filtrate recovered toluene to get hydroxyamitriptyline crude product. After the crude product was refined by recrystallisation with toluene and activated carbon, it was dissolved in concentrated hydrochloric acid under stirring and heating, refluxed for 2-3 hours, and then the dilute hydrochloric acid was evaporated to dryness under reduced pressure. Add isopropyl alcohol and activated carbon, heating and stirring reflux for 1 hour.
Filter while hot, slowly cool the filtrate under stirring, crystallise, filter and dry to obtain amitriptyline hydrochloride. The yield is about 75%.
WHY CHOOES US?

OUR CERTIFICATE

CUSTOM PROCESS

OUR PACKAGE

OUR EXHIBITION

OUR FACTORY

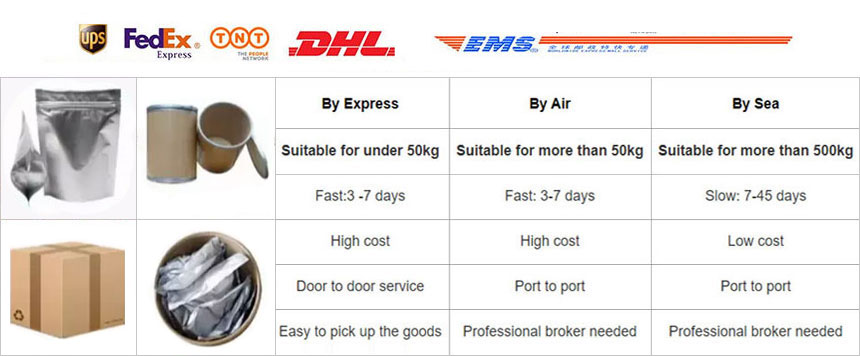
Shipping
Pharmaceutical Intermediate manufacturers
©2022 Xi'an Henrikang Biotech Co., Ltd.,