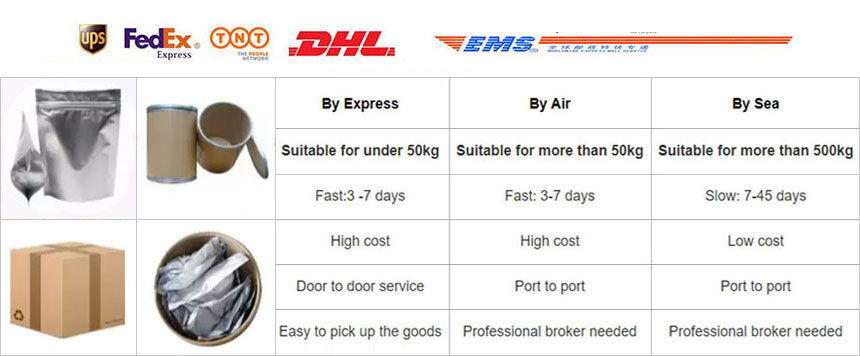
Pharmaceutical
Human APIs Powder
- Respiratory Drugs Raw Material
- Antiviral Antibacterial
- Antipyretic Analgesics
- Antihistamine Drugs
- Antineoplastic
- Cosmetic Raw Material
OEM & ODM
- Capsule
- Softgel Capsule
- Gummies
- Drop & drinks
- Tablets
- Effervescent Tablet
- Gel
- Powder
- Chewing Gum
- Pet Supplement
Veterinary raw materials
Phone: 86-18419147460
E-mail: sales03@nutritionoem.com
Add: Fengcheng 2nd Road, Weiyang District, Xi'an, Shaanxi, China
L(+)-Tartaric acid Raw Materials CAS 87-69-4


Product Overview:
Tartaric acid, 2,3-dihydroxybutanedioic acid. Molecular formula: HOOCCHOHCHCHOHCOOH i.e. dihydroxysuccinic acid. There are two asymmetric carbon atoms and three stereoisomers, i.e., dextrorotatory (D-type, L-type), levorotatory (L-type, D-type), and endorotatory. Usually, the racemic type of tartaric acid is also known as gluconic acid. Dextro-type tartaric acid is widely distributed in higher plants in the form of free or K, Ca, and Mg salts, and is especially found mostly in fruits and leaves. In the manufacture of wine, large quantities of tartaric (potassium hydroxide salt) are deposited. It is also commonly found in molds and lichens.
Product Attributes
CAS:87-69-4
MF:C4H6O6

MW:150.09
EINECS:201-766-0
Specification: 99% min L(+)-Tartaric acid Powder
Sample:L(+)-Tartaric acid Powder
Packaging:1kg/bag, 25kg/drum
Brand: Henrikang
Appearance:White
Storage: Cool Dry Place
Shelf Life: 2 Years
Test Method: HPLC
Product Details
L(+)-Tartaric acid Powder Usage and Synthesis.
Tartaric acid is also an antioxidant with applications in the food industry. It can be utilized as a deoxidizer in biochemical tests. It is also known as 2,3-dihydroxybutanedioic acid.
Structural formula HOOCCH(OH)CH(OH)COOH. Potassium hydrogen tartrate is present in grape juice, this salt is insoluble in water and ethanol, and precipitates in the winemaking process of grape juice, which is called tartar, and the name of tartaric acid is derived from it.
Tartaric acid mainly exists in the form of potassium salts in a variety of plants and fruits, and a small amount exists in a free state.

Uses of L(+)-Tartaric acid.
It is used as an antioxidant added to foods to give them a sour flavor. The largest use of tartaric acid is as a beverage additive. It is also a raw material for the pharmaceutical industry.
In the mirror industry, tartaric acid is an important additive and reducing agent to control the rate of formation of silver mirrors and to obtain very homogeneous coatings.
Used as a biochemical reagent, masking agent and beer foaming agent, also used in the tanning industry.L(+)-Tartaric Acid is widely used as a souring agent for beverages and other foodstuffs in wine, soft drinks, candy, bread, and certain gelatinous sweets. It is mainly used as acidulant, splitting agent and pharmaceutical raw material.
L(+)-Tartaric acid is widely used as an acidulant in beverages and other foodstuffs, for wines, soft drinks, candies, breads, certain gelatinous sweets.
Using its optical activity, as a chemical splitting agent, used in the manufacture of anti-tuberculosis drug intermediate DL-aminobutanol splitting; can also be used as a chiral raw material for the synthesis of tartaric acid derivatives; the use of its acidity, as a polyester fabric resin finishing catalyst, the production of gluten Vitamins PH adjusting agent; the use of its complexation, used as a plating, desulphurization, pickling, as well as chemical analysis, pharmaceutical testing of complexing agent, masking agent, chelating agent, printing and printing. agent, chelating agent, anti-dyeing agent for printing and dyeing; using its reducing properties, used as a reducing agent for chemical mirror making.
Photographic developer. It can also be complexed with many kinds of metal ions, and can be used as cleaning agent and polishing agent for metal surface.

Characteristic of L(+)-Tartaric acid.
Product Method of Bulk L(+)-Tartaric acid Powder.
1. It is obtained by taking the tartar produced in the manufacture of wine as raw material, converting it into calcium salt, and then decomposing it with a slight excess of dilute acid. Or take maleic acid and hydrogen peroxide as raw materials, convert them into epoxysuccinic acid under certain temperature, and then hydrolyze them to get DL-tartaric acid.
It can also be obtained from epoxysuccinic acid by chemical synthesis method, and L(+) tartaric acid by the action of ring-opening enzyme contained in Nocardia succinogenes.
2. Add distilled water to industrial tartaric acid, heat with steam and stir to dissolve. Add an appropriate amount of activated carbon, fully stirred after standing, filtration, filtrate heating and concentration to the surface of the film, while hot filtration, filtrate cooling crystals, to be completely after the crystals with a small amount of distilled water shower at 30 ~ 40 ℃ after drying to the non-staining spoon can be laid flat.
If the control of activated carbon decolorization temperature of 80 ° C, after filtration at 80 ° C concentration under reduced pressure, cooling crystals, the crystals obtained in non-ferrous containers in the recrystallization of refining low-temperature drying, can be obtained dextro-tartaric acid [L (+) tartaric acid] finished products.
Inquire This Product